Ozone depletion: The bullet that missed
The global catastrophe that almost got us
When you have a near-miss experience, you tend to forget it quickly afterward. It’s like driving through an intersection, and a giant SUV barrels through a red light and almost hits you. You may be momentarily shaken up, but a few hours later, you’ve completely forget about how you were almost killed. Such is the nature of our responses to danger once it has passed.
However, let's not be so quick to forget the global catastrophe that nearly claimed us: ozone depletion.
The past
Ozone is a fascinating and crucial component of our atmosphere. While most of the oxygen in the atmosphere is made up of two oxygen atoms bound together (O2), ozone consists of three oxygen atoms (O3). It has the important property of absorbing high-energy ultraviolet (UV) radiation, shielding the Earth's surface from these damaging rays. Such UV radiation is bad for humans as well as plants and animals. In fact, it was the development of an ozone layer in our atmosphere that allowed life to flourish on land.
In the early 1970s, scientists theorized that certain man-made chemicals, known as chlorofluorocarbons or CFCs, had the potential to reduce the amount of ozone in our atmosphere — this became known as ozone depletion. Given the crucial role of ozone in maintaining a livable environment, this caused great concern.
Even before evidence of actual ozone depletion was observed, countries began to take action. For example, the U.S. banned many non-essential uses of the chemicals, such as propellants in aerosol spray cans. This reflected a different view at the time that government should protect its citizens rather than protect the profits of corporations.
By the mid-1980s, the world was busy negotiating the phase-out of the primary ozone-depleting CFCs when the Antarctic ozone hole (AOH) was discovered. The AOH is an annual event: over Antarctica, the majority of the ozone is destroyed during Spring. The ozone builds back up as Spring ends and, by Summer, things are basically back to normal.
The AOH was a stunning discovery. Prior to its discovery, it had been hypothesized that ozone depletion would be relatively slow and occurring at high altitudes over mid-latitudes. In the AOH, however, almost all of the ozone is wiped out in a few weeks. If this happened at mid-latitudes, where people lived, it would be an absolute catastrophe.
The scientific community was initially at a loss to explain the AOH, but by the end of the 1980s the chemistry connecting CFCs to the AOH had been firmly established. The AOH is an example of how uncertainty is not your friend. Things might be better than you think, or things can be much much worse.
In the Montreal Protocol, signed in 1987, the world began the phase out of CFCs. This was another remarkable achievement: a global agreement to phase out an incredibly useful chemical, even without a clear path to how the world would adapt to it.
This gave room for ozone deniers, the 1980s analog to today’s climate deniers, to criticize the agreement. They made basically the same arguments about ozone depletion that are being made today about climate: the science is uncertain, it’s not due to humans, and it’s too expensive to fix.
This is a quote from an article in National Review by Fred Singer, who later became a well-known climate denier. So not only are the arguments the same, but the people are the same.
I think it’s fair to say that all of these arguments have turned out to be wrong. First, the science of ozone depletion has 100% stood the test of time. Second, and most importantly, the economic argument also turned out to be wrong. The phase out of CFCs turned out to be so cheap and easy that almost no one noticed. No one had to give up their air conditioner. We all still have refrigerators that work just as well as the ones in the 1990s.
The parallels with the present climate debate are notable, particularly the claim that we cannot transition to renewable energy without economic catastrophe. Certainly a clean-energy transition won’t be trivial, but modeling from energy centers suggests that we can do it over the next few decades and at low cost — but we need to start now.
The world avoided
My former colleagues at NASA Goddard wrote a paper describing the world we avoided by phasing out CFCs. It paints a grim picture of what the world would look like if the SUV had hit us.
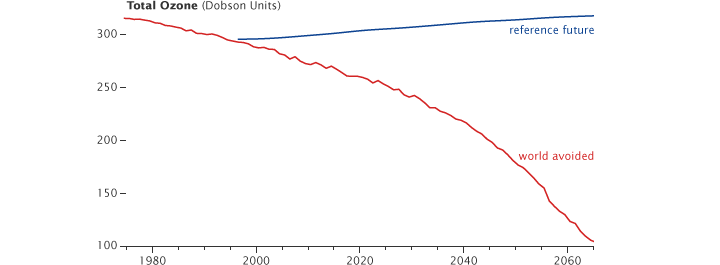
Here is global average ozone:
The reference future is our world, the world avoided is the world that would have existed had we not phased out CFCs. By the 2060s, the world would have lost two-thirds of it’s ozone. This, in turn, would have greatly increased the dangerous ultraviolet radiation reaching the surface. This plot shows the UV dose at noon under clear skies in July in mid-latitudes:
Today’s value of 10 is high risk for UV exposure, which is why public health professionals tell you to wear sunscreen when you go out. The world avoided has a UV index of 30 — three times what is considered high risk and high enough to give you a perceptible sunburn in 5 minutes.
Here is the paper’s lead author, Paul Newman, talking about what would have happened without the phaseout of CFCs under the Montreal Protocol:
The lesson we should not avoid
People do not appreciate how close we came to a real calamity of ozone depletion. I would ask everyone reading this to imagine a grim world with much lower ozone levels and then thank the scientists and policymakers who solved this problem 30 years ago.
But there’s also a lesson here for the future. The ozone problem is similar in may ways to the climate problem and it should serve as a touchstone for how global policy collaboration based on sound scientific research can solve big problems. It also shows how innovation spurred by policy can produce cheap solutions, which is what has happened over the last decade with solar and wind energy. We just need the world’s policymakers to learn the lessons of the bullet that missed.




The window of opportunity for the Montreal Protocol was opened by Richard Benedick who wrote a book about it: „Ozone Diplomacy“.
I don’t think there will be another moment for positive collective action, like we have seen in this example. Getting a country (let alone multiple countries) is like getting a family to agree on pizza toppings for dinner. Thank you for this piece! As a millennial, the ozone was one of the first things I learned about that piqued my interest in climate change.